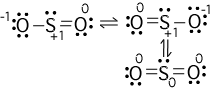
It's bent with a bond angle of 120 degrees. And that's it, the SO2 molecular geometry. Here we can see AX2N is Bent and has a bond angle of 120 degrees. This molecule has a bent geometry, with two oxygen atoms (red) forming a shallow V with a. Lewis Structures & Formal Charge Formal charge is a charge assigned to each atom in a Lewis structure that helps to distinguish among competing structures. Animation of the structure of a molecule of sulfur dioxide (SO2). We can remember that this is a bent molecule, or we can look it up on a table. Valence Shell Electron Pair Repulsion (VSEPR) THEORY & 2. We'll ignore the ones, and we can see that we have AX2N. We only have one unbonded electron pair for SO2. A is the central atom, the Sulfur, we have one of those, X the bonded atoms, those two oxygen atoms, and N that is the unbonded electron pairs. We can also use the AXN notation to determine the shape of SO2. Two double pairs are bonded together and there is one lone pair as well which further gives it a bent shape. The three pairs of bonding electrons lie at an angle of 119 o. Alternatively, the electron geometry of sulphur dioxide is in the shape of a trigonal planar. (b) Because oxygen is more electronegative than sulfur, SO2 has polar bonds. SO 2 molecular geometry is considered to V-shaped or bent. In essence the Valence Shell Electron Pairs are Repelling each other, giving SO2 it's shape. Use the VSEPR model to predict the molecular geometry of (a) O3, (b) SnCl3. Also, a lone pair exists on sulfur atom and each oxygen atom has two lone pairs. The Lewis structure doesn't tell us this directly but if we visualize the atoms and unbonded electron pairs on that central atom, repelling each other, we'll see that oxygen atoms move away from the unbonded pair and give the molecule a bent shape. There are two double bonds between sulfur atom and oxygen atoms in SO molecule. This is because electrons are negative and they repel each other.

First, we can use the Lewis structure for SO2 to visualize how the atoms and unbonded electron pairs will spread out in three dimensions. Although this may be confusing since there are different bonds, a double bond and a single bond is present, including triple bonds of the molecule, it still comes up with the same bond angles on all.We can find the molecular geometry for SO2 it two ways. This means that the o-orbitals are also almost perfect SP2 orbitals. Looking at the molecular geometry of SO2, the bond angle is at 120°. The H-O-H bond angle in H2O is about 109degrees, whereas the bond angle for O-S-O is about 120degrees, EXplain using lewis diagrams why the molecules have the same molecular geometry but different bond angles. There is polarization because Sulfur pulls the charge of the molecule to its side while gaining partial negative charge and not Oxygen since it is the least electronegative atom, making SO2 a polar molecule. The molecular geometries for H2O and SO2 are bent. SO2 has a Bent molecular structure and shape with bond angles of 120. The hybridization of the SO2 is given by sp2. 120 degrees) with asymmetric charge distribution on the central atom. There is also a lone pair above the Sulfur atom. The molecular geometry of SO 2 is bent (approx. Structure Determination by VSEPR There are 4 electron pairs at the corners of a tetrahedron. Most important factor in determining geometry is relative repulsion between electron pairs. In its most stable state, the Sulfur atom forms double bonds with the adjacent oxygen atoms. Rules for making Lewis dot structures Sulfur Dioxide, SO2 SO2 (2) Thiocyanate ion, (SCN)- MOLECULAR GEOMETRY VSEPR Valence Shell Electron Pair Repulsion theory. There is an imbalance charge across other atoms in the molecule, making Sulfur Dioxide polar. SO2 comprises a Sulfur atom surrounded by two oxygen atoms. A quick explanation of the molecular geometry of SO4 2- including a description of the SO4 2- bond angles.Looking at the SO4 2- Lewis structure we can see th. With SO2, the pairs of bonding electrons are arranged at an angle of 120 degrees. Electron geometry includes all the pairs of electrons, even the lone pairs. This is not similar to molecular geometry, where only the total number of atoms is considered to determine its shape. Molecular geometry is the 3-dimensional shape that a molecule occupies in space This is the molecular geometry of CF4 Quite the same Wikipedia It is an orange solid that is soluble in water Korean Drama Gangster Falls In Love NOCl: nitrosyl chloride NOCl: nitrosyl chloride. The electron geometry of SO2 is trigonal planar.
